AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
 
In this model n = ∞ corresponds to the level where the energy holding the electron and the nucleus together is zero. For instance, hydrogen is the simplest atom and thus, it has the simplest spectrum.\) is the Rydberg constant in terms of energy, Z is the atom is the atomic number, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. 
The frequencies of the light emitted by a particular element exhibit a regular pattern.Emission Line spectrum is when an atomic gas or vapor is excited at low pressure by passing an electric current through it, the emitted radiation has a spectrum that contains specific wavelengths only.The atomic spectra can be defined as the spectrum of the electromagnetic radiation that is emitted or absorbed by electrons during transitions between different energy levels within an atom. I also found all the wavelengths of the other solutions.Rutherford’s discovery of the nucleus of the atom in 1911 answered many questions concerning the structure of the atom. Their cause, however, remained unexplained until the structure of the atom and, in particular, its electronic structure, was solved. The first Atomic structure model was proposed by J. The phenomenon of atomic spectra has been known since the mid-1800s.Important Notes On Atoms Important Questions The empirical formulas are given for the observed wavelengths, series such as Lyman, Paschen, Brackett and Pfund and Balmer formula in terms of frequency of light are important as the wavelengths give that hydrogen atom radiate or absorb but these results are empirical and don’t give any reasoning as to why only certain frequencies are observed in the hydrogen spectrum.The Balmer formula when written in terms of frequency of light is.Here are several line spectra: 5.3 Electron Levels, Sublevels, and Orbitals Bohr’s model could not explain the emission spectra for all the elements. Atomic structure and spectra 2.1 Atomic structure 2.1.1 The hydrogen atom and one-electron atoms TheHamiltonianforone-electronatomssuchasH,He ,Li2 .,canbewrittenas H p2 2me Ze2 40r, (2.1) where p is the momentum operator, me is the electron mass (me 9.10938291(40)× 1031kg), Z is the atomic number (or proton number), e is. The Lyman series is in the ultraviolet region while Paschen and Brackett's series are found in the infrared region. Each atom has its own unique line spectrum this is virtually an 'atomic fingerprint' that can be used identify the element. 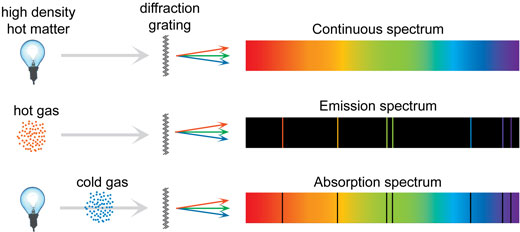
There are other series of spectra for hydrogen which was discovered after their discoverers such as Lyman, Paschen, Brackett, and Pfund series, and these are represented by the given formulae:.Beyond this limit, no further distinct lines appear, and instead, only a faint continuous spectrum is seen. When n = ∞ is considered, one obtains the limit of series at λ = 364.6 nm and this is the shortest wavelength in the entire Balmer series.This is also known as the Balmer formula. In the above formula, λ is the wavelength while R is known as the Rydberg constant and n can have integral values like 3, 4, 5, etc. 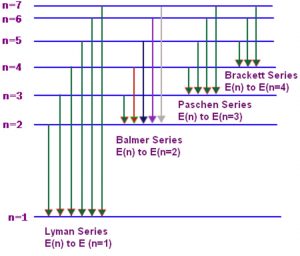
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
